Images
Participantes

Contact

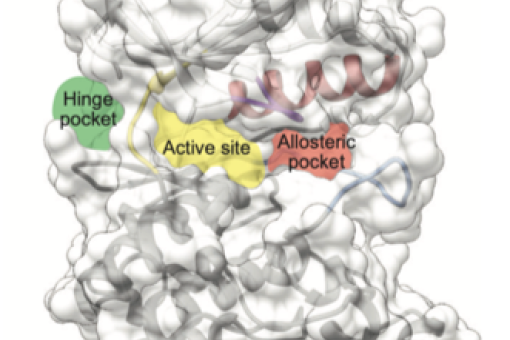
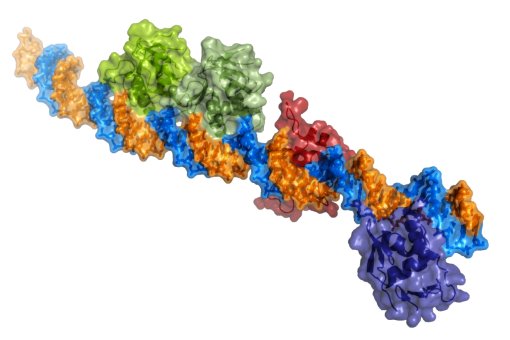
BMC ha publicado un artículo sobre el “Cancer Genome interpreter” (CGI), una plataforma abierta diseñada para ayudar en la identificación de las alteraciones tumorales que llevan al desarrollo del cáncer y detectar aquellas que puedan ser un objetivo terapéutico. Núria López-Bigas, líder del laboratorio de Genómica Biomédica del IRB Barcelona, forma parte de esta iniciativa.
Enlace a BMC
IRB Barcelona
El Instituto de Investigación Biomédica (IRB Barcelona) trabaja para conseguir una vida libre de enfermedades. Desarrolla una investigación multidisciplinar de excelencia para curar el cáncer y otras enfermedades vinculadas al envejecimiento. Establece colaboraciones con la industria farmacéutica y los principales hospitales para hacer llegar los resultados de la investigación a la sociedad, a través de la transferencia de tecnología, y realiza diferentes iniciativas de divulgación científica para mantener un diálogo abierto con la ciudadanía. El IRB Barcelona es un centro internacional que acoge alrededor de 400 científicos de más de 30 nacionalidades. Reconocido como Centro de Excelencia Severo Ochoa desde 2011, es un centro CERCA y miembro del Barcelona Institute of Science and Technology (BIST).