Organizado por IRB Barcelona en colaboración con la Fundació Catalunya La Pedrera.
Presentation

Objectivos
El programa Crazy About Biomedicine es un curso dirigido a estudiantes de primero de bachillerato que deseen explorar algunos de los descubrimientos fascinantes que se están haciendo actualmente en las ciencias de la vida. A través de este curso, los estudiantes tendrán la oportunidad de profundizar su conocimiento de la teoría y técnicas científicas en el campo de la biomedicina. Trabajarán junto con investigadores jóvenes para experimentar cómo se hace ciencia en un instituto de investigación internacional, ganar un poco de experiencia práctica en las últimas metodologías de vanguardia y posicionarse para una posible carrera profesional en las ciencias de la vida.
Descripción del curso
Taller de un año de duración sobre las ciencias de la vida para estudiantes de bachillerato. Organizado por el IRB Barcelona dentro del Programa "Bojos per la Ciència" de la Fundació Catalunya La Pedrera.
Este curso combina sesiones teóricas y actividades experimentales prácticas, que se llevarán a cabo durante 16 sábados del año. El curso tratará 10 temas científicos actuales, que van desde la biología celular y molecular hasta la biología estructural y computacional y la química, presentados por investigadores jóvenes del IRB Barcelona. En el primer semestre (enero-abril), los tres primeros sábados se dedicarán a estas sesiones teóricas generales para todos los participantes. Durante los seis sábados siguientes, se formarán grupos pequeños que entrarán a los laboratorios para las sesiones prácticas. A continuación, se repetirá este programa con 5 temas de investigación nuevos para el segundo semestre (mayo-noviembre). Los estudiantes participantes deberán comprometerse a asistir al curso durante todo el año.
Los estudiantes recibirán un certificado de participación al finalizar el curso en una ceremonia especial de clausura, en la que podrán asistir padres y profesores.

Idioma del curso
Todas las charlas y sesiones prácticas se harán en inglés.

Fechas y horarios
El curso tendrá lugar de enero a noviembre de 2022, 10.00-14.00h.
1r SEMESTRE
- Sáb. 14 enero 2022: Inauguración oficial del curso
- Sáb. 22 enero: Bienvenida y charla 1
- Sáb. 5 febrero: Charlas 2-3
- Sáb. 19 febrero: Charlas 4-5
- Sáb. 26 febrero: Sesión práctica 1
- Sáb. 12 marzo: Sesión práctica 2
- Sáb. 26 marzo: Sesión práctica 3
- Sáb. 2 abril: Sesión práctica 4
- Sáb. 30 abril: Sesión práctica 5
2º SEMESTRE
- Sáb. 21 mayo: Charla 1
- Sáb. 4 junio: Charlas 2-3
- Sáb. 11 junio: Charlas 4-5
- Sáb. 17 septiembre: Sesión práctica 1
- Sáb. 1 octubre: Sesión práctica 2
- Sáb. 15 octubre: Sesión práctica 3
- Sáb. 22 octubre: Sesión práctica 4
- Sáb. 5 noviembre: Sesión práctica 5
Precio del curso
Los participants tendrá que abonar a la Fundació Catalunya La Pedrera la cantidad de 425 euros. Se pueden consultar la oferta de becas en su página web.
Sitio donde se realizará el curso
Institut de Recerca Biomèdica (IRB Barcelona)
C/ Baldiri Reixac, 10
08028 Barcelona
Quién puede solicitar una plaza
Este curso está dirigido a los estudiantes de primero de bachillerato que tengan un interés y talento especiales en los campos relacionados con las ciencias de la vida.
Los estudiantes pueden solicitar plaza a un máximo de 3 de los programas de la serie "Bojos per la Ciencia" y finalmente sólo podrán participar en uno de ellos.
Cómo solicitar una plaza
Las inscripciones se tendrán que realizar aquí a partir del 15 de septiembre 2021.
Los estudiantes interesados tendrán que rellenar el formulario de solicitud y incluir una carta de motivación. También se pedirá una carta de recomendación directamente de dos de sus profesores que conozcan bien el alumno. En el caso de que el estudiante haya cambiado de centro este curso, sugerimos que soliciten las cartas a los antiguos profesores.
La fecha límite de inscripción es el 25 de octubre 2021 (23:59h).
El curs está abierto a un total de 25 estudiantes. Se seleccionaran los candidatos en función de su expediente académico, de les recomendaciones de sus profesores y de su motivación para participar. Se invitará a los candidatos preseleccionados a hacer entrevistas con los organizadores científicos en noviembre, para después hacer la selección final. La primera semana de diciembre se comunicará el resultado a los estudiantes. Se pedirá a los estudiantes seleccionados y a sus padres/tutores legales que firmen una carta de compromiso que asistirán a todas las sesiones.
Colaboradores
Facebook: @LaPedrera.Fundacio
Twitter: @PedreraFundacio
Instagram: @lapedrera_fundacio
Facebook: @LaPedrera.Ciencia
Twitter: @PedreraScience
Instagram: @lapedrera_scienceacademy

Para cualquier duda, por favor contactad-nos a: irb_outreach@irbbarcelona.org
Fechas importantes
- 25 octubre 2021: Fecha límite inscripción
- 12 noviembre 2021: Contacto con los candidatos pre seleccionados
- 13-28 noviembre 2021: Entrevistas
- 30 noviembre 2021: Contacto con los candidatos seleccionados
- 14 enero 2022: Inauguración oficial del curso
- 22 enero 2022: Inicio del curso
- Acto de Clausura - fecha exacta a concretar
Programme
SEMESTER 1
1. Unravelling the Molecular Structure of Life
Blazej Baginski (Structural Characterization of Macromolecular Assemblies)

Proteins and nucleic acids (DNA and RNA) are the basic building blocks of life. They perform a multitude of functions–from sensing, transporting, and enzymatic regulation, to building the cell’s internal skeleton. Therefore, the fold and 3D-structure of these biomolecules is carefully controlled. A protein’s 3D structure determines its activity, creates receptor binding pockets and enzyme active centres.
Crystallography is one of the few methods that allows the structural determination of such macromolecules with atomic precision. By studying the interactions of crystallised molecules by means of high energy X-rays, it is possible to pinpoint the location of atoms and their bonds in a given molecule of interest.
During this course, we will set up a protein crystallisation experiment, learn the operation of high-precision pipetting robots, and cryogenically freeze protein crystals to prepare them for X-ray data collection at the synchrotron.
2. Manipulating cellular plasticity
Isabel Calvo (Cellular Plasticity and Disease)

The concept of cellular plasticity has gained great relevance during the last years in the context of cancer and tissue repair. Cellular plasticity allows adult cells to regress to stem cell-like states through de-differentiation pathways.
It is possible to convert differentiated cells into pluripotent stem cells (induced pluripotent stem cells or iPSCs) by the simple expression of four transcription factors. These iPSCs are functionally equivalent to embryonic stem cells (ESCs), which are derived from the developing blastocyst and can divide indefinitely while maintaining the capacity to differentiate into any cell type of the organism.
There is an increasing interest in better understanding how these transitions occur both in vitro and in vivo and how they can be manipulated. This knowledge will be directly applied in regenerative medicine to improve current medical treatments.
Students will learn the basic techniques to culture differentiated and pluripotent stem cells and they will be trained to induce the transition between cellular states. Finally, they will have the opportunity to perform in vitro assays that will help us to recognize the ultimate state of pluripotency.
3. CRISPR WARS: The Gene Strikes Back
Luis Povoas (Gene Translation Laboratory) 
Some years ago, during the process of constructing a fly model to study human diseases our lab found a very interesting protein that seems to be responsible for many different processes in the cell. This protein is a derivate of a group of proteins responsible to introduce amino acids in tRNA, to enable protein synthesis. We called it SLIMP (seryl-tRNA synthetase-like insect mitochondrial protein). Past studies suggest that this new protein has acquired an essential function in insects, being one of the most interesting results obtained so far, the role of SLIMP in cell cycle progression. To uncover more of SLIMP’s functions, our lab is using a revolutionary technology that changed the way we make science. This technology is the CRISPR editing system. This tool enables us to precisely edit the genome of live cells. With it, we can remove genes (knock-out), introduce genes (knock-in), introduce markers to follow a specific protein (tags) and others. Therefore, with this technology, we expect to uncover SLIMP intracellular location, protein-protein interactions and try to understand the SLIMP knockdown phenotypes in relation to DNA damage and cell cycle checkpoints. During this workshop, you will be introduced to concepts of molecular cloning, cell culture and CRISPR technology. May the science be with you!
4. From biomedicine to computational biology
Alba Sala (Molecular Modelling and Bioinformatics)

The interaction between small molecules is essential to living organisms. Amongst these molecules we find protein and nucleic acids, whose interaction plays a crucial role in biology (e.g., in the storage, repair, expression and regulation of genetic information).
Protein-DNA interaction is mostly defined by two mechanisms: what is called a direct or base read out and an indirect or shape readout. The former describes the ability of proteins to distinguish different binding modes. The later, focuses on the importance of certain protein and DNA sequences to adopt different 3D structures which determine the binding sites. The amount of importance given to these two mechanisms varies from protein to protein, but it is crucial to understand the role of a protein’s 3D structure.
In this course we will study the 3D structure of different proteins from a computational point of view. A protein’s structure is believed to dictate its function, and once a protein’s shape is understood, its role within the essential biological mechanisms can be determined; giving then information to scientists to develop new methodologies that work with a protein’s shape.
5. Introduction to Drosophila melanogaster research: a versatile model in biology & medicine
Bitarka Bisai (Development and Morphogenesis in Drosophila)
 The fruit fly, Drosophila melanogaster, is used as a model organism to study disciplines ranging from fundamental genetics to the development of tissues and organs. Despite the obvious differences between humans and Drosophila, it is remarkable that the fruit fly shares many similarities and conserved pathways with humans. Drosophila genome is 60% homologous to that of humans, less redundant, and about 75% of the genes responsible for human diseases have homologs in flies.
The fruit fly, Drosophila melanogaster, is used as a model organism to study disciplines ranging from fundamental genetics to the development of tissues and organs. Despite the obvious differences between humans and Drosophila, it is remarkable that the fruit fly shares many similarities and conserved pathways with humans. Drosophila genome is 60% homologous to that of humans, less redundant, and about 75% of the genes responsible for human diseases have homologs in flies.
Multipotency has been the focus of extensive work in biomedicine research. However, we lack much knowledge of multipotent cells in their physiological context in the full organism. During this workshop, we will address some of these issues by exploiting a naturally occurring population of multipotent cells, the adult progenitor cells (APCs) in Drosophila. In Drosophila, most larval cells, mainly polyploid, die at the transition between larval and adult stages; only the APCs survive and proceed into their terminal differentiation during metamorphosis. However, both APCs and larval cells are affected by the same nutritional and hormonal cues, suggesting that unique molecular components act in the adult progenitor cells to differentially regulate the effect of the external and the intrinsic stimuli in their unique setting.
This course provides hands-on experience of different techniques to understand Drosophila melanogaster research. You will be introduced to first steps in fly genetics through
experimentation by crossing flies, identifying genetic markers and balancer chromosomes, and applying other genetic tools used every day in the lab. We will learn about the different stages of development and the anatomy of the fly at different stages. We will also perform fly dissections and use advanced microscopy.
SEMESTER 2
1. Strategies to Understand Aging and Senescence
Marta Kovatcheva & Valentina Ramponi (Cellular Plasticity and Disease)


Scientific research and modern medicine have dramatically extended life expectancy, with the average person in the developed world expected to reach 80 years of age or more. However, this extension in lifespan has had no effect on health span; that is, the number of healthy years a human lives. Aging is still characterised by multiple pathologies including frailty, heart disease, cancer, and neurodegenerative diseases, among many others.
One of the main hallmarks of ageing is cellular senescence, the phenomenon by which normal cells stop dividing. Senescent cells accumulate in an organism over time, secreting pro-inflammatory molecules and contributing to age-related diseases. There is an increasing interest in clinical medicine to better identify and target senescent cells, as their elimination may delay and ameliorate some age-associated diseases.
This course provides hands-on experience using different techniques to induce cellular senescence in normal cells. We will learn state-of-the-art techniques to study the molecular biology of senescent cells, analysing in vitro and in vivo samples. Finally, we will perform classical protocols, such as SAβgal staining, to detect senescent cells. These techniques will help us to understand, identify and target senescent cells for clearance, which is a promising therapeutic approach to extend health span.
2. Mirror, Mirror on the Wall… Who Is the Most Helpful Insect of Them All? – Getting to Know Tumorigenesis Through the Fly
Elena Fusari & Amanda González (Development and Growth Control Laboratory)
The fruit fly Drosophila melanogaster has been widely used as a model organism for more than one hundred years to address biological questions in various fields. This organism has emerged as a potent tool for genetic manipulation, offering innumerable possibilities to analyze the detailed interaction between cells and tissues.
One question could be raised: how can a tiny organism such as Drosophila melanogaster be used to understand the pathways of such a complex disease as cancer? Well, the answer is as simple as that: many biological mechanisms are well conserved across evolution, including the ones concerning pathological conditions. This is what allows scientists to translate the knowledge acquired from less complex organisms to more complex ones. For instance, 75% of human disease-related genes and 68% of human cancer-related genes have a counterpart in the fly. We are more similar to the fly than what we may think!
During this semester, we will learn how to study the complex process of tumorigenesis using this model organism, both with a local and systemic approach. We will focus mainly on carcinomas, the most common type of tumor diagnosed in humans.
Carcinomas are derived from epithelial tissue, such as the skin, and they can become invasive or metastatic by spreading beyond the primary tissue layer and surrounding tissues or organs. In aggressive cancer cells, this transition is mediated by the activation of the EMT (Epithelial to Mesenchymal Transition) program, which causes the cells to undergo morphogenetic alterations that increase invasive capacity.
In this course, we will introduce you to the first steps in fly genetics. We will cross flies, identify genetic markers and balancer chromosomes, and apply other genetic tools that we use in the lab every day in order to generate tumorigenic flies and study them. We will also learn about the anatomy of the fly in adult and larvae stages and use advanced microscopy to see several genetic markers in the tumoral tissues. We will also perform in vivo dissections and their respective immunohistochemistry.
3. NMR to study molecules in solution
Miriam Condeminas (Structural Characterization of Macromolecular Assemblies)
Structural Biology aims at understanding the shape and function of biological molecules to decipher the basic mechanisms of life. It can also help identify molecules with pharmacological applications to treat human diseases. Nowadays, such questions can be answered using a wide variety of complementary tools that include Nuclear Magnetic Resonance (NMR).
In NMR, samples are irradiated with radio waves while placed inside a strong magnetic field. As a result, frequency changes in the nuclei of the molecules can be recorded and the corresponding signals plotted into NMR spectra. By combining various types of NMR experiments, each signal can be linked to a particular nucleus and thus yield important information regarding its chemical environment. Given that these experiments can be performed in aqueous solutions, NMR is very versatile in that it enables the observation of macromolecular flexible regions, as well as dynamic processes (invisible using other techniques) and it can also map binding interfaces.
In this course, students will learn the fundamentals behind NMR, how to interpret the spectra resulting from various experiments and the workflow required for assigning a peptide.
4. An omics complex puzzle? Let’s decipher it by using Bioinformatics!
Olfat Khannous (Molecular Modelling and Bioinformatics) & Marina Murillo (Gene Translation Laboratory)

 Complex biological diseases such as cancer and Alzheimer are caused by a combination of genetic and environmental factors, and in many cases we have not yet identified or established a relationship between them to shed light to the understanding of the origin and progression of such biological problems.
Complex biological diseases such as cancer and Alzheimer are caused by a combination of genetic and environmental factors, and in many cases we have not yet identified or established a relationship between them to shed light to the understanding of the origin and progression of such biological problems.
Multi-omics profiling and integration combines different types of data for a better understanding of these complex diseases.
One of the branches of the ‘Omics’ sciences is transcriptomics which involves the study of all the RNA molecules (including mRNA, tRNA, rRNA, and other non-coding RNAs) within a cell, otherwise known as the transcriptome. Consequently, by analyzing the transcriptome researchers can determine if a gene is turned on or off in the cells and tissues of an organism.
On the other hand, metagenomics is the study of the genetic material (genomes) from a mixed community of organisms of a sample. It allows us to describe which microbial communities are living within each individual (microbiome) and its relationship with host genetics, diet and environment can reveal links with disease or health.
In our workshop you will be immersed in the computational world (dry lab) understanding its synergy with the experimental lab (wet lab), and you will learn how to obtain transcriptomics and metagenomics data and also how to use bioinformatics tools to analyze them and to add missing pieces to these mentioned complex puzzles.
5. CRISPR/Cas9 to generate a KO
Camilla Bertani (Translational Control of Cell Cycle and Differentiation)

My laboratory is focused on studying a family of proteins called Cytoplasmic Polyadenylation Element Binding Proteins (CPEBs), are sequence-specific mRNA-binding proteins that control translation in development, health and diseases such as cancers. To evaluate the role these proteins, play in cancer cells, a good approach is to induce KO of the protein and evaluate the resulting phenotype.
For this reason, in the laboratory we have designed a possible genomic editing approach, based on the CRIPR / Cas9 technique. Our aim is to induce the deletion of an exon present in the genomic sequence of these proteins, in order to block their functionality.
Therefore, before being able to evaluate the phenotype, it is necessary to evaluate which of the clones we have obtained really presents the deletion of the exon and also, to evaluate if in homozygous or heterozygous form.
For this reason, it is essential to conduct the screening of clones, in particular the analysis of genomic DNA, through the use of a technique called PCR.
.
Venue
El programa Crazy About Biomedicine se llevará a cabo a las instalaciones del IRB Barcelona.
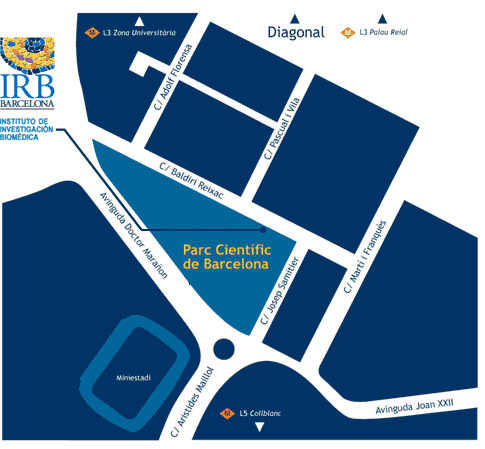
Institute for Research in Biomedicine (IRB Barcelona)
Parc Científic de Barcelona
C/ Baldiri Reixac, 10
08028 Barcelona

