Professor (IBMB-CSIC)
Research information
Background
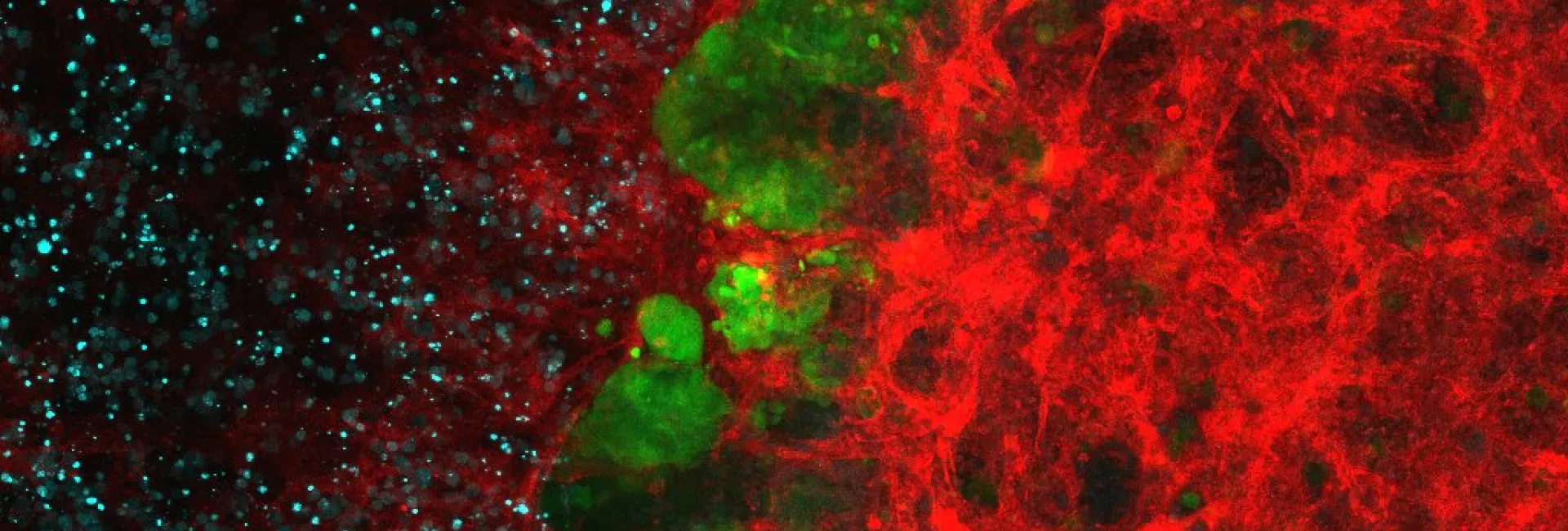
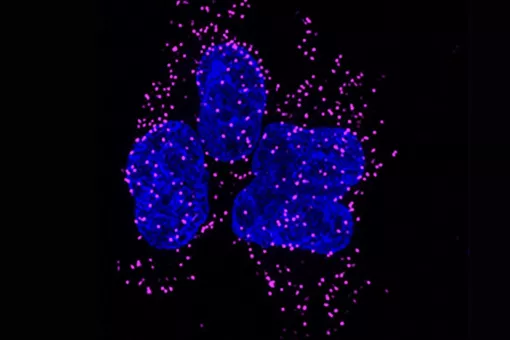
Studies on the genetic control of development have proven to be very been fruitful in unveiling the mechanisms impaired in many disease conditions. This is particularly true in the case of cancer and associated illnesses, as can be assessed by the wealth of knowledge obtained from the study of the developmental role of oncogenes and tumour suppressor genes, the studies of cell migration, epithelial-mesenchymal transition and vasculogenesis. In this regard, the focus of our research at the IRB is to advance in providing basic knowledge on the genetic regulation of such developmental processes, and in particular in their control by cell communication mechanisms. Our aim is to study those phenomena in the context of the whole organism and to do so we use Drosophila melanogaster as a tool. The choice of Drosophila as a model system to study the basic phenomena in development has proved to be extremely fruitful. The broad conservation of the developmental key genes identified in Drosophila and the basic similarity between developmental processes in different species justifies the choice of an organism as a model system.
Research interests
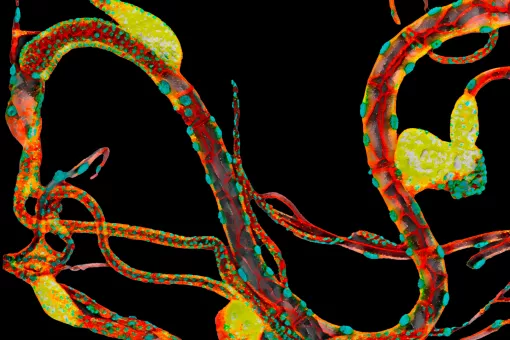
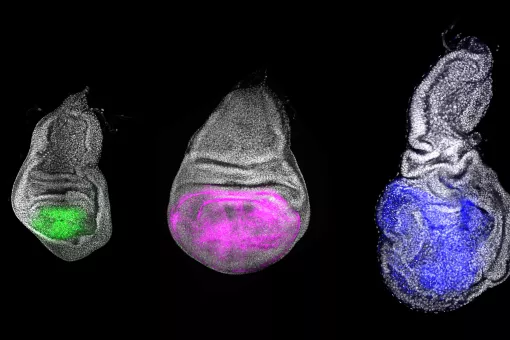
The work of many laboratories has allowed to begin to understand the genetics logic behind development and we are now trying to understand how these mechanisms impinge on cell behaviour. In particular, we put our emphasis on the issue of the supracellular organisation and more precisely on how individual cell changes can generate an upper level of complexity such the one found in tissues and organs. We work in this research field using two Drosophila models already well known in the lab (the transduction pathway specifying the embryonic terminal regions and the tracheal system), as well as another one more recently added (the morphogenesis of the endoderm and gut). We hope that a global approach using these three systems will allow us unveil how transcription factors and signalling pathways trigger and coordinate changes in cell issues such as shape, polarity and adhesion into collective organization and migration. In performing this global approach we also consider an evolutionary perspective that could account for the recruitment of already existing mechanisms into distinct processes or for their particular redeployment to explain appearance of different kinds of supracellular organization. Finally, we hope this approach will be also crucial to provide an understanding of some of the mechanisms involved in disease conditions and in particular in oncogenesis. To this aim, we have fostered our collaboration with some research groups from the oncology programme.
Research lines
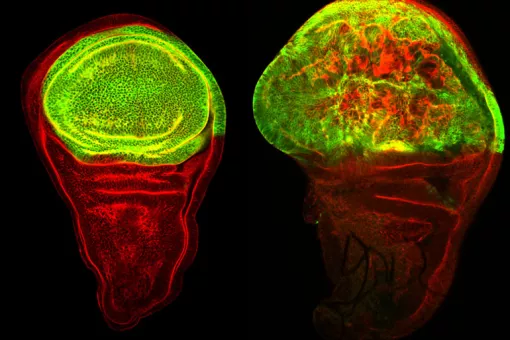
- Drosophila Adult Progenitor Cells as a paradigm of cell plasticity, multipotency and reprogramming in vivo
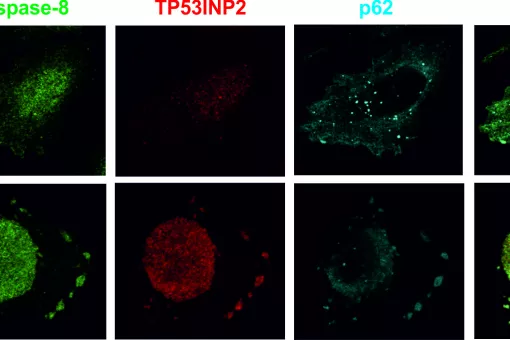
- Cell survival and cell death in metamorphosis
- Differential gene expression in diploid vs polyploid cells
- The role of headcase in progenitor cells