Images
Researchers in Barcelona discover more potential candidates on the route to tailored, photo-switchable therapies by disproving design limitation
Last year, a group of scientists at the Insititute for Bioengineering of Catalonia (IBEC), IRB Barcelona and the University of Barcelona announced that they had achieved photo-switchable, or light-regulated, molecules to control protein-protein interactions – key determinants in biological processes, and therefore highly promising therapeutic targets – in a remote and non-invasive manner. The discovery signified a major step towards the development of targeted, light-controlled drugs, whose effects would be limited to a specific area and time, reducing side effects.
Now, in recent experiments, the same researchers have been using their photo-sensitive molecules to control clathrin-mediated endocytosis, a process whereby cells absorb certain substances that they need to function properly. In doing so, they’ve made a discovery that greatly expands the field of potential inhibitors, as it questions the need for one of the previously supposed design requisites of these molecules.
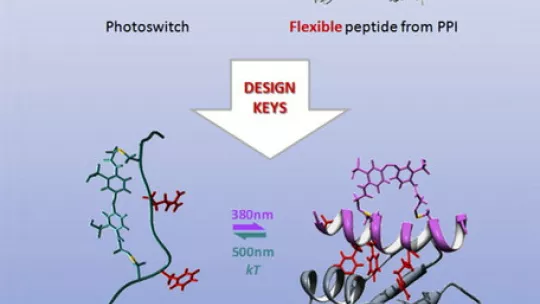
“As many protein-protein interactions are mediated by short, often helical, linear peptides (small proteins), we thought our photoswitchable molecules had to be designed to mimic these rigid peptides in order to be able inhibit the PPI,” explains Pau Gorostiza of IBEC’s Nanoprobes and Nanoswitches group. “In fact, we found that they don’t need to have the same rigid structure, and that flexible structures actually show a greater inhibitory capacity, as well as better photoswitching ability.”
The absence of a rigid, helical structure, therefore, is not a limitation when selecting candidates for photoswitchable inhibitors of PPIs, meaning that when developing further potential inhibitors, researchers can look to the wide group of more flexible peptides for their candidates.
"These results are another step forward a general approach to photocontrol therapeutically relevant intracellular protein-protein interactions," says Ernest Giralt, leader the Peptides and Proteins group at IRB Barcelona. "The behavior showed by these peptides highlights the advantage that can proceed from the partial flexibility of some protein-protein interaction inhibitors, which allows the binder to structurally ‘adapt’ to the target protein," adds Laura Nevola, co-first author of the paper at IRB Barcelona.
The most immediate therapeutic applications expected for photoswitchable inhibitors is for diseases affecting superficial tissue such as the skin, retina and external mucus membranes. In the future, photo-sensitive molecules could be used to study, for example, in vitro endocytosis in cancer cells – where this process is uncontrolled – which would allow selective inhibition of the cells’ proliferation.
Another area of application would be in developmental biology, where cells require endocytosis to change shape and function. Their development also opens up entire new fields of research, such as optopharmacology and optogenetics.
--
Reference article: André Martin-Quiro, Laura Nevola, Kay Eckelt, Sergio Madurga, Pau Gorostiza and Ernest Giralt (2015). “Absence of a Stable Secondary Structure is not a Limitation for Photoswitchable Inhibitors of b-Arrestin/b-Adaptin 2 Protein-Protein Interaction”. Chemistry & Biology, pub ahead of print. DOI: http://dx.doi.org/10.1016/j.chembiol.2014.10.022
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).