Images
Maria Macias, Pau Martín, and Joan Massagué publish a featured review article in Trends in Biochemical Sciences (TIBS) on the Smad protein family, the members of which play a key role in cancer.
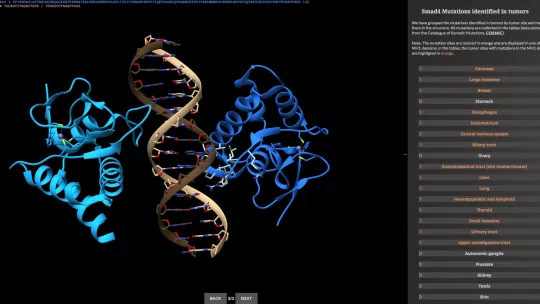
The article is accompanied by a web application in which available 3D structures of Smads can be explored, as can their alterations in several metazoa, their variability in humans, and their mutations in tumours.
Maria Macias, ICREA researcher at IRB Barcelona and head of the Structural Characterization of Macromolecular Assemblies Lab, and Joan Massagué, director of the Sloan Kettering Cancer Center, have published a review article on a web application on Smad proteins in the June edition of Trends in Biochemical Sciences (TIBS), part of the Cell group.
Smad proteins comprise a family of transcription factors that transmit the signals produced by TGF-Beta hormones, which are fundamental for embryonic development, tissue regeneration, and correct cell function. In addition to these activities, Smad proteins attract interest because of their tumour suppression capacity. Mutations in Smads limit this capacity, thus leading to the accumulation of these proteins in tumours. “Current data indicate that more than 25% of pancreatic and colon tumours have mutations in Smad4 and Smad2. Most of these mutations are involved in the loss of the function of these proteins, which could be correlated with alterations in interaction regions and with damage in the 3D structure,” explains Macias.
The article covers information available on Smad sequences and analyses how variations affect the structure, regulation and function of these proteins. “The objective is to take advantage of the information generated by genome sequencing (of organisms, human populations and tumours) and to combine it with the protein structures available. “ For the first time, we have access to sequences not only of model organisms but also of enigmatic ones such as placozoa, numerous vertebrates, including Neanderthal humans and present-day primates, and thousands of sequences from human populations, as well as variations in several kinds of tumours. The idea is to examine whether there are common patterns of differences and to analyse their possible effect on the structure and function of Smad proteins,” says Macias.
3D imaging
The amount of information gathered from the diverse genomic databases was so great that the scientists decided to build a webapp, to facilitate the comparison of Smad sequences and to show the 3D structures of the proteins and the mutations associated with disease and to publish it in the article, together with more conventional figures. “This idea was welcomed by the editor of TIBS,” comment the researchers.
“Smad proteins show extraordinarily high conservation throughout evolution, thereby indicating that they are key components for life”. “In general, they are mutated in many tumours in very distinct tissues. Through this review and web application, the mutations in the structures can be easily appreciated,” explains Macias.
The article provides a complete structural snapshot of distinct kinds of Smads and contributes to visualizing how mutations affect their structure and function—knowledge that could be exploited for therapeutic purposes in the future.
Web app: http://smad.maciasnmr.net/
Reference article:
Structural determinants of Smad function in TGF-β signaling
Maria J. Macias, Pau Martín-Malpartida and Joan Massagué
Trends in Biochemical Sciences (June 2015) DOI: http://dx.doi.org/10.1016/j.tibs.2015.03.012
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).