Images
Ángel R. Nebreda’s team (IRB Barcelona) publishes a study in the journal Nature Communications addressing the role of the p38 protein in angiogenesis—the formation of new blood vessels—a critical process that fuels tumour cells and allows them to grow and eventually develop metastases.
A greater understanding of how new blood vessel formation is regulated could help to improve chemotherapy treatments for cancer, as well as to develop more efficient angiogenic therapies for other diseases.
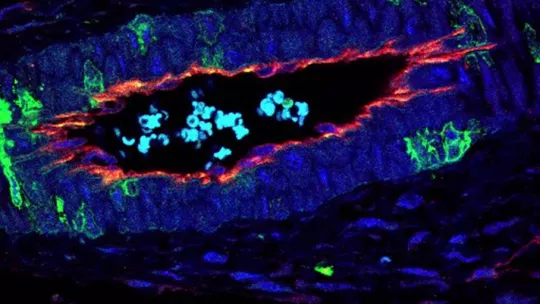
A study performed at the Institute for Research in Biomedicine (IRB Barcelona) and published in the journal Nature Communications demonstrates that inhibition of the p38 protein boosts the formation of blood vessels in human and mice colon cancers. Known as angiogenesis, this process is critical in fuelling cancer cells, allowing them to grow and to eventually develop metastases.
The team led by Ángel R. Nebreda, head of the Signalling and Cell Cycle Laboratory at IRB Barcelona, has shown that the activity of p38 is important in mesenchymal stem cells (MSCs), which show high plasticity and can localize around blood vessels. These cells participate in various processes, such as tumour development, and this study unveils their relevance in the regulation of angiogenesis.
The activity of p38 in cancer cells had already been described. However, until now, little was known about the role of this protein in MSCs and even less about its involvement in angiogenesis.
The objective of this study was to examine the role of p38 in new blood vessel formation during tumorigenesis, in particular considering the contribution of MSCs.
In this regard, as explained by Nebreda, ICREA researcher at IRB Barcelona, the study demonstrates that “p38 represses angiogenesis by exerting its action specifically in MSCs. Using genetic mouse models, we show that the inhibition of p38 stimulates the formation of new blood vessels both in tumours and during the repair of damaged tissue”.
The conclusions of this study shed light on the mechanisms that regulate blood vessel formation and may have implications for the optimisation of chemotherapy-based cancer treatments, as well as for the treatment of diseases involving compromised angiogenesis.
“We hope that the biological knowledge that our work has generated can contribute to the development of more efficient therapies,” concludes Raquel Batlle, postdoctoral fellow at IRB Barcelona and first author of the study.
The research has been funded by the European Research Council, the Fundación Olga Torres, the Marató de TV3, the Ministry of Science, Innovation and Universities, and AGAUR (Agency for Management of University and Research Grants).
Reference article:
Raquel Batlle, Eva Andrés, Lorena Gonzalez, Elisabet Llonch, Ana Igea, Núria Gutierrez-Prat, Antoni Berenguer-Llergo and Angel R. Nebreda
Regulation of tumor angiogenesis and mesenchymal-endothelial transition by p38α through TGF-β and JNK signaling
Nature Communications (2019) DOI: 10.1038/s41467-019-10946-y
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).