Images
This breakthrough is crucial as recent studies demonstrate that there is a correlation between the severity of these diseases and the intermediates formed during the process of amyloid fibril formation.
Researchers at the Institute for Research in Biomedicine (IRB Barcelona), in collaboration with the University of Cambridge, have designed a new strategy to obtain structural insights into the nature of different species populated during the process of amyloid fibril formation linked to diseases such as Alzheimer’s and Parkinson’s. The study, published in the journal Proceedings of the National Academy of Sciences (PNAS), describes a combination of biophysical techniques and methodologies that has allowed to gain both mechanistic information on the process of amyloid fibril formation and structural information on the different species present during the course of aggregation.
Natàlia Carulla, ICREA researcher at IRB Barcelona, who works in Ernest Giralt’s laboratory in the Chemistry and Molecular Pharmacology Programme, and first author of the paper, explains that "intermediate species populated during amyloid fibril formation are so relevant and there is so little known about them that it is very important to have tools to study them in detail".
The aggregation of certain proteins into amyloid fibrils is associated with increasingly prevalent disorders such as Alzheimer´s disease and Parkinson´s disease. Thus, for example, in the brains of individuals with Alzheimer’s, the beta amyloid protein aggregates into amyloid fibrils that accumulate in deposits known as plaques. This is the reason why initial studies proposed that beta amyloid fibrils caused the disease. Later, it was observed that there were only weak correlations between levels of amyloid fibrils in the brain and the severity of dementia. This finding led to the formulation of new hypotheses centred on the role of intermediates, namely stages of aggregation formed before the development of fibrils. "There is no consensus about the type of intermediate responsible for the disease but it is known that there is a greater correlation between intermediates and the degree of cognitive deficit in patients" explains the researcher.
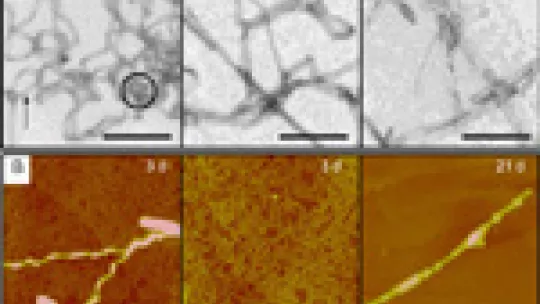
These researchers have applied this new protocol to the study of the aggregation process of a model protein, an SH3 domain, which forms fibrils but is not associated to any disease. The strategy includes labelling the protein with pulses of deuterated water, water holding deuterium, that is to say heavy hydrogen, and mass spectrometry and nuclear magnetic resonance analyses. By applying this protocol at different times during the aggregation process, the researchers have obtained information about the regions of the aggregates that are least accessible to the solvent and therefore involved in aggregation.
Carulla goes on to explain that in Giralt’s lab, they will now apply this protocol to study the intermediates formed during beta amyloid protein aggregation into amyloid fibrils, which is associated with Alzheimer’s. One of the group’s goals is to test the mechanism of action of certain molecules that inhibit fibril development, some of which are currently in clinical trials. In addition, identification of the protein regions involved in aggregation will allow the design of therapeutic molecules that fit into these regions and prevent the formation of toxic intermediates.
Reference article:
Experimental characterization of disordered and ordered aggregates populated during the process of amyloid fibril formation
Natàlia Carulla, Min Zhou, Muriel Arimon, Margarida Gairí, Ernest Giralt, Carol V. Robinson, and Christopher M. Dobson
PNAS 2009; DOI 10.1073/pnas.0812227106
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).