Images
For the scientific community, the protein POP (prolyl oligopeptidase) is an attractive therapeutic target for the treatment of cognitive and neurodegenerative disorders.
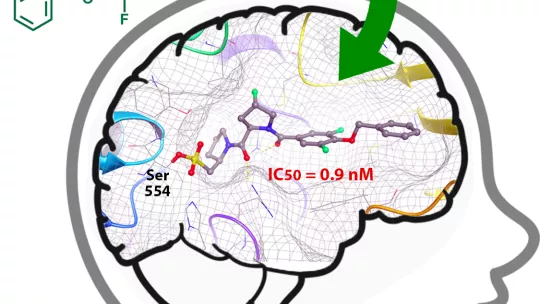
Scientists at IRB Barcelona have developed the first POP inhibitors that are irreversible, selective and brain-permeable.
An article published in Cell Chemical Biology by scientists at the Institute for Research in Biomedicine (IRB Barcelona) describes a new class of prolyl oligopeptidase (POP) inhibitors. Found mostly in the brain, POP has emerged as an “attractive·” therapeutic target for the treatment of several cognitive and neurodegenerative disorders, such as Parkinson’s disease and schizophrenia.
Most POP inhibitors developed to date have a short half-life in blood, do not show sufficient selectivity for the protein, and do not cross the blood-brain barrier (which protects the brain), thus limiting their potential as drugs.
Work done by IRB Barcelona postdoctoral fellow Salvador Guardiola describes the first class of POP inhibitor characterised by long half-life, selectivity and capacity to enter the brain. The Design, Synthesis and Structure of Peptides and Proteins lab, headed by Ernest Giralt, has generated and characterised a family of 15 modified peptides (small proteins), called peptidomimetics, which have shown promising results.
"Natural peptides are not stable in tissues and have relatively weak activity. In contrast, if we modify them with synthetic amino acids and introduce functional groups designed to react with the therapeutic target, we can achieve peptides that are much more active and efficient," explains Guardiola.
At low concentrations, these new peptides inhibit POP in a rapid, specific and sustained manner, both in the isolated enzyme and in human intact cells. In addition, they are highly selective for two proteases that are similar to POP (DPPIV and FAP) and show a high capacity to cross the blood-brain barrier, say the researchers.
"These inhibitors pave the way for the development of new treatments for diseases of the central nervous system and will also contribute to studies on the role of POP in these disorders and to the development of inhibitors of other proteases," explains Giralt, who is also a senior professor at the University of Barcelona (UB).
This study has been funded by the Ministry of Economy and Competitiveness, ERDFs, and the Catalan government. It has involved the UB’s Nuclear Magnetic Resonance Unit, IRB Barcelona’s Mass Spectrometry and Proteomics Facility, and the Barcelona Supercomputing Center. The work was done in collaboration with scientists at the University of Glasgow (Scotland, UK), the Utrecht Institute for Pharmaceutical Sciences (the Netherlands,) and the biotech Iproteos, a spin-off from IRB Barcelona and the UB.
More about Ernest Giralt's research: "The power of the medicinal chemistry" - Meet Our Scientists
Reference article:
Salvador Guardiola, Roger Prades, Laura Mendieta, Arwin J. Brouwer, Jelle Streefkerk, Laura Nevola, Teresa Tarragó, Rob M. J. Liskamp, Ernest Giralt
Targeted covalent inhibition of prolyl oligopeptidase (POP): discovery of sulfonylfluoride peptidomimetics
Cell Chemical Biology (2018) DOI: 10.1016/j.chembiol.2018.04.013
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).