Images
- A group of scientists headed by Maria Macias, ICREA researcher at IRB Barcelona, describes the mechanism through which TGIF1 inhibits the activity of Smad proteins.
- The study, which used molecular and structural biology techniques to analyse this interaction, has been published in Nucleic Acids.
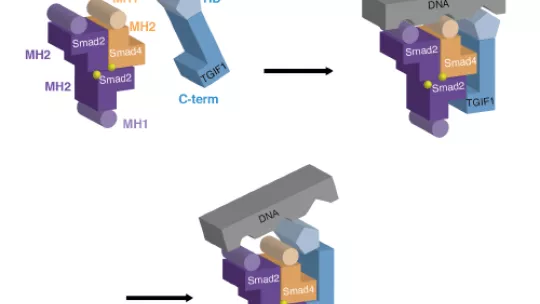
A complete understanding of the mechanism of interaction between Smad proteins (Smads)—involved in many aspects of cell development—and their main inhibitor, the protein TGIF1, paves the way for the design of specific molecules for the treatment of a wide spectrum of conditions and alterations of normal body function. The Structural Characterization of Macromolecular Assemblies lab at the Institute for Research in Biomedicine (IRB Barcelona) has described this mechanism of interaction and confirmed that TGIF1 inhibits Smads by occupying the DNA binding region. Without binding to DNA, Smads are unable to exert their action as transcription factors, thus blocking the TGF-β signalling pathway.
A virtually universal signalling pathway
Signalling pathways are formed by a group of biomolecules that work together to regulate one or more cell functions. In such a pathway, a molecule activates and/or interacts with others, which in turn activate others, and this goes on successively to form what is known as a cascade effect.
Smads form part of the signalling pathway orchestrated by the TGF-β family of hormone, which is conserved in all multicellular animals. Smads make up a family of transcription factors that activate and inhibit gene transcription, thus influencing key processes such as embryonic development, tissue regeneration and immune response. The TGIF1 protein is one of the main factors known to inhibit Smads and, when mutated, it is associated with holoprosencephaly (a serious congenital malformation of the brain and face), as well as tumour progression in several kinds of cancer.
"We believe that our study will be of great interest to many groups worldwide who are conducting research into the impact of TGF-β signalling in cancer and other diseases," says Maria Macias.
A synergistic interaction
The interaction between Smads and TGIF1 had been proposed in numerous previous studies based on biochemical and cell experiments. The team of scientists headed by Macias has now used molecular and structural biology techniques such as X-rays and nuclear magnetic resonance to study the fine details of the interaction.
This study has confirmed that Smads have multiple interaction sites that can act in a synergistic manner, thus increasing the complementarity and specificity of the interactions. Another conclusion drawn from the study is that the regions previously through to bind exclusively to DNA also participate in interactions with proteins and cofactors. “The identification of Smad binding sites for cofactors would unveil the presence of new target regions that could be used for therapeutic purposes,” explains Maria Macias.
The study has involved the Unit on Asymmetric Synthesis at IRB Barcelona and has been supported by competitive funding from the Ministry of Economy and Competitiveness (now Ministry of Science, Innovation and Universities), BioMedTec (run by the Obra Social ‘la Caixa’), the BBVA Foundation, CERCA, and ICREA. It was also supported by a predoctoral ‘la Caixa’ grant and a posdoctoral Marie Sklodowska-Curie COFUND grant.
Reference article:
Ewelina Guca, David Suñol, Lidia Ruiz, Agnieszka Konkol, Jorge Cordero, Carles Torner, Eric Aragon, Pau Martin-Malpartida, Antoni Riera and Maria J. Macias
TGIF1 homeodomain interacts with Smad MH1 domain and represses TGF- β signaling
Nucleic Acids Research (2018) DOI: 10.1093/nar/gky680
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).