Images
For several decades the pharmaceutical industry has been interested in achieving peptide analogues —similar molecules with improved properties— of the natural hormone somatostatin. This hormone and another two analogue compounds, octreotide and lanreotide, are used for the treatment of several kinds of neoplasia, such cancer of the thyroid, pancreas and prostate, in diseases associated with the growth hormone, for example in gigantism, and in digestive hemorrhages. Headed by Antoni Riera (University of Barcelona) and Maria Macias (ICREA Research Professor), a team of researchers at IRB Barcelona, in collaboration with the company BCN Peptides, has designed a peptide that is ten times more stable in blood than the natural hormone and is more active than the two analogues currently available on the market. The success of the design lies in the substitution of two sequences in the natural sequence for two non-natural amino acids. Angewandte Chemie, the second highest ranking journal in chemistry, has published the article in the advanced online edition and has selected it for the inside cover in the first issue of February.
“The use of the natural hormone has two disadvantages: it degrades rapidly in the organism (2-3 minutes) and it has a wide spectrum of activities. Thus when attempting to correct certain diseases without affecting the normal function activated by the natural hormone, it is important to develop compounds that show greater stability in blood and that are more selective in their action. What we have obtained in the lab is a peptide that fulfills these two objectives”, explains Antoni Riera, head of the Asymmetric Synthesis Laboratory at IRB Barcelona, where the new compound has been synthesized. “Also, it could be developed into a drug rapidly because this type of molecule does not usually cause toxicity”, the scientist adds.
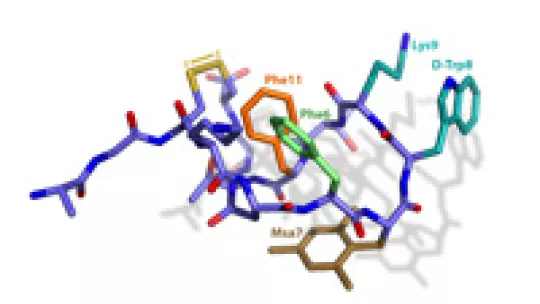
The study performed by Drs. Riera and Macias also provides for the first time structural data on the many conformations of the natural hormone somatostatin. The hormone adapts its structure depending on the function that it performs, which is determined by its interaction with distinct receptors (five receptors have been characterized to date). These multiple conformations make it impossible to obtain a single structure for somatostatin either in solution or using X-ray diffraction techniques. “Through the resolution of the structure of the compounds synthesized and comparison with data on the natural hormone”, explains Maria Macias, head of the Protein NMR Spectroscopy Laboratory, “we have unraveled one of the active conformations of somatostatin in its interactions with one of the receptors, specifically SSTR2”. For the authors of the study, it has been both the innovative chemical synthesis of the molecule and the new structural approach combined with functional assays that have allowed a more rational design of analogues of the natural hormone. According to these researchers, this study opens the way for further designs of analogues that would be selective in their interaction with other receptors.
The first author of the article is Pablo Martín–Gago, a PhD student at IRB Barcelona, and the study has involved ten other researchers, among these three from the company BCN Peptides and three from the University of Alcalá de Henares.
Reference article
Fine-tuning the π-π Aromatic Interactions in Peptides: Somatostatin Analogues Containing Mesityl Alanine.
Martín-Gago P, Gomez-Caminals M, Ramón R, Verdaguer X, Martin-Malpartida P, Aragón E, Fernández-Carneado J, Ponsati B, López-Ruiz P, Cortes MA, Colás B, Macias MJ, Riera A.
Angew Chem Int Ed Engl. 2012 Jan 5. doi: 10.1002/anie.201108928. [Epub ahead of print]
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).