Images
Genomic instability is the main risk factor for tumour development in humans. Therefore understanding its origin and exploring therapeutic targets is paramount.
Histone 1 silences a region of the genome that causes irreparable DNA damage when translated and is lethal for the organism.
Scientists headed by Ferran Azorín, CSIC research professor and head of the Chromatin Structure and Function group at the Institute for Research in Biomedicine (IRB Barcelona), have discovered why histone 1 is a major protection factor against genomic instability and a vital protein for the organism. Devoted to the function of histone 1, the least known of the five histones, the study has been published today in the journal Nature Communications.
"Although histone 1 is key component of chromatin –the form in which DNA is packaged inside the cell nucleus through the action of histones -, there are still many questions open regarding this molecule," says Ferran Azorín. "Regarding the other histones, which are major proteins in the regulation of gene expression, we know which enzymes modify them, their functions, and how they are regulated... But for some reason, the functions of histone 1 have not been addressed," he adds.
Lethal lesions
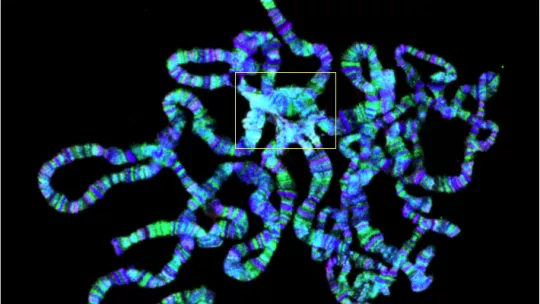
The study explains for the first time that the suppression of histone 1 causes cell damage and genomic instability (DNA damage). The deregulation of a commonly suppressed region of chromatin, called heterochromatin, leads to defects in information transcription, which in turn gives rise to the accumulation of DNA and RNA hybrids, the so-called R-loops, which are lethal.
"The deregulation of heterochromatin has disastrous consequences," explains Jordi Bernués, associate researcher in Azorín’s group and coleader of the study. Furthermore, the team also observed that, in the presence of histone 1, these problems did not arise in spite of heterochromatin expression. "Histone 1 not only serves as a repressor but also actively contributes to the removal of R-loops". However, the researchers do not know how this function comes about. "The mechanism is what we want to study, how histone 1 prevents the mechanism from causing damage," explains IRB Barcelona PhD student Anna Casas-Lamesa, co-first author of the article together with Aleix Bayona-Feliu.
The fruit fly Drosophila melanogaster has been crucial in unravelling this function. First, only one variant of this histone is present in the fly—while in humans there are up to seven—so the research is simplified. Second, the fly allows scientists to remove a protein from a specific place and at a specific time. They removed histone 1 from the precursor structure of wings. They observed that the fly was born alive but without wings; so the removal of this molecule caused the death of all cell precursors of this tissue (if histone 1 is removed from the entire fly, the embryo dies).
A statistical analysis of gene expression allowed the scientists to reject a long-standing hypothesis, namely that histone 1 is a global repressor of expression. "The effect of removing histone 1 on gene expression is very weak," says Bernués. Its expression alters that of “only” 5% of genes. "It is not a transcriptional regulator," confirms Azorín.
Heterochromatin contains genetic information that is not translated for proteins but that comprises repetitive sequences that the cell silences and that is tightly regulated. "Both R-loops and the information held in heterochromatin have natural functions but when they are deregulated they are lethal. "We have now been able to associate genomic instability with the uncontrolled formation of R-loops caused by the lack of histone 1 and this is completely new," emphasised Bernués.
Histone 1 and cancer
Preliminary experiments in cultured tumour cells confirm that the genomic instability present in these cells is partly caused by histone 1 deficiency. "But we have to further study the functions of this histone, identify the other proteins with which it is associated, and determine the enzymes that modify it and why and the cell signalling pathways involved," explain the scientists.
In addition to exploring the mechanisms through which histone 1 keeps heterochromatin in check and prevents R-loop formation, Anna Casas-Lamesa is also addressing the involvement of this molecule in cancer. "Our preliminary studies are promising," explains Azorín "but we are still a long way off proposing histone 1 as a target".
This study has been supported by the Ministry of Economy and Competitiveness, through ERDFs, and by the Government of Catalonia.
Reference article:
Aleix Bayona-Feliu, Anna Casas-Lamesa, Oscar Reina, Jordi Bernués and Fernando Azorín
Linker histone H1 prevents R-loop accumulation and genome instability in heterochromatin
Nature Communications (2017) doi: 10.1038/s41467-017-00338-5
About IRB Barcelona
Created in 2005 by the Generalitat de Catalunya (Government of Catalonia) and University of Barcelona, IRB Barcelona is a Severo Ochoa Centre of Excellence, a seal that was awarded in 2011. The institute is devoted to conducting research of excellence in biomedicine and to transferring results to clinical practice, thus improving people’s quality of life, while simultaneously promoting the training of outstanding researchers, technology transfer, and public communication of science. Its 25 laboratories and seven core facilities address basic questions in biology and are orientated to diseases such as cancer, metastasis, Alzheimer’s, diabetes, and rare conditions. IRB Barcelona is an international centre that hosts 400 employees and 32 nationalities. It is located in the Barcelona Science Park. IRB Barcelona forms part of the Barcelona Institute of Science and Technology (BIST) and the “Xarxa de Centres de Recerca de Catalunya” (CERCA).
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).