Images
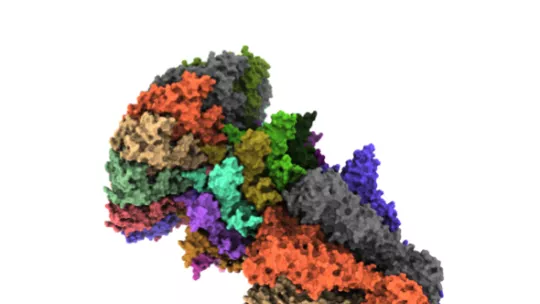
The study reveals the opening and closing mechanism of the portal protein during the maturation of the viral capsid, the structure that carries the genetic material of the virus.
The researchers have combined cryomicroscopy and crystallography techniques to study these viruses that infect bacteria.
A study involving the Spanish National Research Council (CSIC) and the Institute for Research in Biomedicine (IRB Barcelona) has deciphered the structure and mechanism of action of a key complex in infection by the bacteriophage T7 virus. The results have been published in the journal Nature Communications.
The work is the result of the collaboration of experts in electronic cryomicroscopy of the National Centre for Biotechnology (CSIC) led by José L. Carrascosa and in X-ray crystallography of the Institute of Molecular Biology of Barcelona (CSIC) and IRB Barcelona directed by Miquel Coll. By combining the results of the two techniques, the research groups have obtained new data on the mechanism of action of this complex in the process of viral maturation.
“Bacteriophages are viruses that infect bacteria. In addition to their interest as a model, because of their genetic simplicity and high structural complexity, they are now a new focus of attention for researchers because of their potential as an alternative to the use of antibiotics,” explains José L. Carrascosa, a scientist at the National Centre for Biotechnology and co-director of the study.
A viral capsid is formed during the cycle of bacteriophage T7. This structure stores the genetic material of the virus, introducing it through a small entrance channel formed by the portal protein and that is subsequently closed by the virus's tail protein. The mechanism that allowed this “door” to open and close in a controlled manner was unknown until now.
“These bacterial viruses have a DNA packaging mechanism similar to that of herpes viruses that infect humans. Therefore, deciphering how they work helps us to understand how pathogenic viruses affect us,” says Miquel Coll, a researcher at the Institute of Molecular Biology of Barcelona and IRB Barcelona and co-director of the study.
First authors of the work, Ana Cuervo, from the National Centre for Biotechnology, and Montserrat Fàbrega-Ferrer, from IRB Barcelona and the Molecular Biology Institute of Barcelona, highlight how “combining electronic cryomicroscopy and synchrotron X-ray crystallography has allowed us not to only to solve the atomic structure of a large complex, which is a technical challenge, but also to define a model of portal opening and closing during the maturation of the viral capsid”. In this way, the scientists have been able to identify the interactions between the different proteins that allow changes in the conformation of the complex, from an open structure to allow the passage of viral DNA, to a closed one when forming mature viral particles.
This work has been funded by the “Severo Ochoa” and the “María de Maeztu” Programmes of excellence and the “Ramón y Cajal” Programme run by the Ministry of Science, Innovation and Universities, and the European iNEXT and Instruct-ERIC projects.
Reference article:
Ana Cuervo, Montserrat Fàbrega-Ferrer, Cristina Machón, José Javier Conesa, Francisco J. Javier Fernández, Rosa Pérez-Luque, Mar Pérez-Ruiz, Joan Pous, María Cristina Vega, José L. Carrascosa y Miquel Coll.
Structures of T7 bacteriophage portal and tail suggest a viral DNA retention and ejection mechanism.
Nature Communications (2019) DOI: 10.1038/s41467-019-11705-9
Source: CSIC
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).