Images
- Researchers led by Dr. Xavier Salvatella have uncovered the mechanism by which an experimental drug is able to block intrinsically disordered proteins, which were previously considered “undruggable”.
- Published in Science Advances, the study shows that the drug does not act on the protein in isolation, but instead takes advantage of the moment when they briefly group together into small complexes (oligomers).
- This discovery forms the scientific foundation for the IRB Barcelona spin-off Nuage Therapeutics and will enable the design and optimization of treatments for cancer and other diseases associated with these types of proteins.
Intrinsically disordered proteins lack a fixed structure, which is why they have been considered "undruggable" targets for drug development for years. However, these proteins play a key role in numerous diseases—ranging from various types of cancer to neurodegenerative disorders—long limiting the therapeutic options available to treat them.
Now, a team from IRB Barcelona led by Dr. Xavier Salvatella has successfully explained how a drug is able to bind to one of these disordered proteins, revealing a mechanism that opens new avenues for therapeutic design. The study, published in the journal Science Advances, shows that the key lies not in the protein in isolation, but in a transient state that occurs when multiple copies of the protein begin to cluster.
“For a long time, these proteins were thought to be impossible to target because they lack a stable structure. Our study demonstrates that, at certain moments, they adopt more organized conformations, and these windows of opportunity can be exploited to develop new treatments,” explains Dr. Xavier Salvatella, ICREA researcher and head of the Molecular Biophysics Laboratory at IRB Barcelona.
A moving target, but not an impossible one
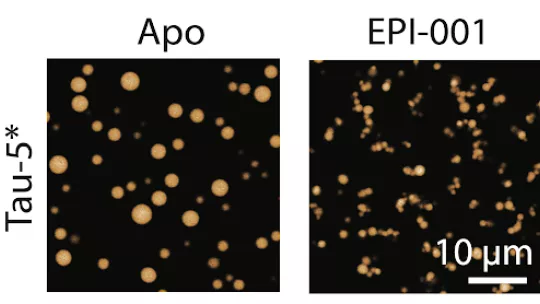
Intrinsically disordered proteins play a key role in many cellular processes, but because they lack a rigid, well-defined structure, they have been considered impossible targets for drug development for years. This new study demonstrates that this view was incomplete: when these proteins begin to cluster, they form transient oligomers in which certain structural elements appear. It is in this intermediate state that the drug can recognize its target and bind to it selectively, as the team has shown using biophysical techniques and cellular experiments.
Understanding the mechanism to design better drugs
“Understanding the mechanism is a key step; it allows us to optimize these molecules and design new ones that take advantage of these transient states in disordered proteins,” adds Dr. Stasė Bielskutė-García, the first author of the study.
This knowledge not only explains why this compound works, but also provides a rational basis for designing new drugs targeting these types of proteins—opening the door to new therapeutic strategies for diseases that currently have limited treatment options.
The scientific foundation of Nuage Therapeutics
The mechanism described in this study provides the scientific foundation for the IRB Barcelona spin-off Nuage Therapeutics, which was created to develop drugs targeting intrinsically disordered proteins—a class of therapeutic targets traditionally considered inaccessible.
The company applies this knowledge to identify and optimize molecules capable of recognizing the transient structural states of these proteins, following the exact strategy now detailed in the publication. Currently, Nuage Therapeutics focuses its efforts on small-cell lung cancer and various gastrointestinal cancers—two areas with significant unmet medical needs where these types of proteins play a key role.
Beyond oncology, Nuage Therapeutic’s ultimate goal is to become the leader in the field of intrinsically disordered proteins, expanding the use of this technology into therapeutic areas where protein disorder plays a key role and treatment options remain limited.
The study published in Science Advances thus reinforces the scientific foundation of the company’s approach and illustrates how fundamental research conducted at IRB Barcelona can be transformed into new therapeutic opportunities.
The work involved collaboration with Dr. Denes Hnisz’s group at the Max Planck Institute for Molecular Genetics (Germany), researchers from the University of Florence (Italy), and Dr. Antoni Riera, also from IRB Barcelona.
The research received funding from the Spanish Ministry of Science, Innovation and Universities, the Agency for Management of University and Research Grants (AGAUR), ”la Caixa” Foundation, the Spanish Association Against Cancer (AECC), the Mark Foundation for Cancer Research, and the European Research Council (ERC).
Related article:
Oligomerization enables the selective targeting of an intrinsically disordered region by a small molecule
Stasė Bielskutė-García, Borja Mateos, Muhammad Awawdy, Carla Garcia-Cabau, Henri Niskanen, Carolina Sánchez-Zarzalejo, Lorenzo Bracaglia, Roberta Pierattelli, Isabella C. Felli, Marta Frigolé-Vivas, Jesús García, Antoni Riera, Denes Hnisz, Xavier Salvatella
Science Advances (2026) DOI: 10.1126/sciadv.adz74
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).