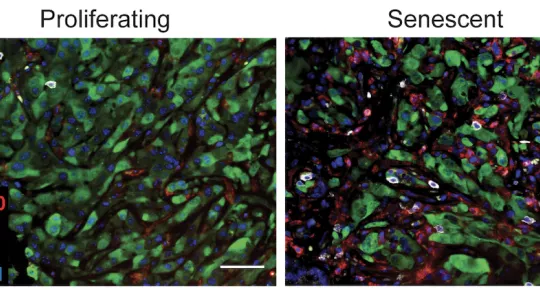
Images
- Senescence amplifies the response of tumour cells to external signals, such as interferon, making them more “visible” to the immune system.
- Published in the journal Cancer Discovery, the finding paves the way for harnessing senescence to enhance anti-tumour immunity and the effectiveness of immunotherapy treatments.
Senescence is a state in which cells do not proliferate but do not die either and, instead, begin to produce and secrete proinflammatory factors. It is induced by diverse cellular stresses, such as upon activation of some oncogenes (cancer-related genes) or in response to anti-cancer drugs. When this happens, senescence can drive potent tumour suppressive responses by alerting the immune system of the presence of mutated or damaged cells or contributing to the efficacy of some therapies. However, senescent cells are not always recognised and/or effectively eliminated by the immune system for reasons that are still not well understood. In this scenario, their sustained presence in tissues creates an inflamed environment that can promote cancer progression and other inflammation-associated diseases. In this context, drugs targeting senescent cells or enhancing their immune-mediated elimination are being explored as possible treatments for cancer and other inflammation-associated disorders.
A study led by Dr. Scott W. Lowe from Memorial Sloan Kettering Cancer Center (MSK) (New York, USA) and Dr. Direna Alonso-Curbelo, now head of the Inflammation, Tissue Plasticity and Cancer laboratory at IRB Barcelona, has discovered a new feature of senescent cells that makes them more “visible” to the immune system: a rewired capacity to sense extracellular signals, such as interferon, that enhances their immunogenicity and can potentiate the anti-tumour activity of the immune system. The work has been carried out using innovative murine models of liver cancer that recapitulate tumour and immune response features relevant to patients' outcomes.
Published in the February issue of Cancer Discovery, the work focused on describing how the induction of senescence in tumour cells reshapes the communication between the tumour and the immune system. "Until now, most studies have focused on the ability of senescent cells to 'send' inflammatory signals to their environment. Our work demonstrates that this communication is bidirectional, and it reveals that senescence increases the capacity of cells to “receive” signals from their environment. These signals activate key pathways for cell recognition and destruction by cytotoxic T cells," explains Dr. Alonso- Curbelo.
Reconnection of an immunogenic “sensing” programme
According to the mechanism discovered by the researchers, the induction of senescence launches a cellular programme that not only remodels the tissue environment but also increases the receptivity/sensitivity of senescent cells to specific environmental signals. Stimulating this senescence-associated “sensing” programme can make cancer cells more visible to the body's immune system, and it is necessary to produce effective immune responses against cancer cells.
"We envision that investigating this and other tissue remodelling and sensing programmes in pre- and post-treatment tumour biopsies may reveal new response biomarkers and/or combination strategies to improve the clinical management of cancer," explains Dr. Lowe, Chair of the Cancer Biology and Genetics Program at MSK’s Sloan Kettering Institute.
The study was first authored by Dr. Hsuan-An (Sean) Chen, a graduate student from Louis V. Gerstner Jr. Graduate School of Biomedical Sciences at MSK and now a postdoc fellow at Rockefeller University, and involved researchers from MSK, Cold Spring Harbor, Calico Life Sciences, and IRB Barcelona. In Dr. Alonso-Curbelo’s lab at IRB Barcelona, co-author Dr. Katharia Woess recently received a Postdoctoral EMBO grant to study the alteration of tissue sensing programmes in the context of tumour development.
A parallel study supports the conclusions
This work has been published together with a discovery from the Cellular Plasticity and Disease Laboratory at IRB Barcelona, which reached complementary conclusions using a very different approach. This parallel study, first-authored by Inés Marín and led by ICREA researcher Dr. Manuel Serrano and Federico Pietrocola from the Karolinska Institutet (Stockholm, Sweden) details how inducing senescence improves the effectiveness of the anti-tumour immune response more than the dead cells regularly used for this purpose.
In this regard, these scientists have verified that vaccination with senescent cells significantly reduces cancer outgrowth in experimental models by eliciting anti-tumour cytotoxic T-cell responses.
Related article:
Senescence rewires microenvironment sensing to facilitate anti-tumour immunity
Hsuan-An Chen, Yu-Jui Ho, Riccardo Mezzadra, Jose M. Adrover, Ryan Smolkin, Changyu Zhu, Katharina Woess, Nicholas Bernstein, Georgia Schmitt, Linda Fong, Wei Luan, Alexandra Wuest, Sha Tian, Xiang Li, Caroline Broderick, Ronald C. Hendrickson, Mikala Egeblad, Zhenghao Chen, Direna Alonso-Curbelo & Scott W. Lowe
Cancer Discovery (2022) DOI: 10.1158/2159-8290.CD-22-0528
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).