Images
In EMBO Molecular Medicine, IRB Barcelona scientists report a technique based on a combination of CRISPR/Cas9 genome editing and patient-derived tumour organoids that allows the study of cell heterogeneity in human tumours.
The novel approach was used to examine the behaviour of colorectal cancer stem cells by tracing specific marker genes.
An innovative methodology proposed by researchers with the Colorectal Cancer Laboratory at IRB Barcelona opens up the possibility to study human tumours by means of tracing genetic modifications. Such an approach had previously been feasible only in animal models and it may have broad applicability for the analysis of cancer cell diversity during human tumour development in vitro or after transplantation of tumours to a recipient animal.
“There is a large interest to study how distinct tumour cell population contribute to the evolution of the disease. Until know, these studies have necessary involved experiments of tumour dissociation and cell isolation, which have raised many conceptual and technical caveats. In essence, this new approach brings the power of mouse genetic tools to study human cancer and overcomes the limitations of identifying and tracking tumour cell populations in an intact environment,” explains ICREA Research Professor and CIBERONC member, Eduard Batlle, who led the study published today in EMBO Molecular Medicine.
The researchers combine two novel methodologies, namely patient-derived tumour organoids (an organoid is a miniaturized and simplified version of an organ that is produced in vitro in three dimensions and that shows realistic micro-anatomy), and CRISPR/Cas9 genome-editing techniques (which allow the introduction of markers into genes of interest). The scientists consider that the combined method proved “particularly well suited” for analysing the diversity of cell populations within cancers as it allows the labelling and tracing of distinct tumour cells through desired marker genes.
“For the first time we can analyse cell lineage relationships in growing tumours, and this technique will help us to address how distinct cell populations contribute to growth, dissemination, and resistance to therapy,” wrote the IRB Barcelona authors in reference to the impact expected.
Tracking colorectal cancer stem cells
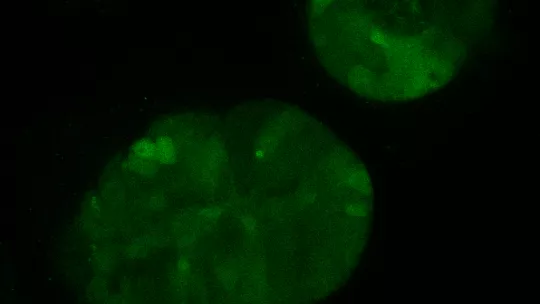
To illustrate the usefulness of this approach, postdoctoral fellow Carme Cortina and PhD student Gemma Turon, who contributed equally as first authors of the study, engineered human colorectal cancer cell organoids to carry a fluorescent marker inserted at LGR5 genes locus, which encodes a stem cell-specific gene. Until now, analysis of cells that express this gene in human tumours had been hampered by the lack of good commercial reagents to recognise this protein. “We have been able to follow LGR5+ cancer stem cell gene for the first time and reveal novel information about their behaviour in human cancers,” says Carme Cortina.
The analysis demonstrated that LGR5+ cells express a gene programme similar to that of normal intestinal stem cells and that they are capable of propagating the disease to recipient hosts very efficiently. The experiments also confirmed that human colorectal cancer cells adopt a hierarchical organisation reminiscent of that of the normal colonic epithelium.
Furthermore, Batlle and colleagues implemented this technology to map the fate of these cells within human CRC and discovered that LGR5+ tumour cells show long-term renewal and multilineage differentiation capacity—both hallmarks of stemness. Finally, the scientists also demonstrated that a subset of LGR5+ cells are dormant and do not divide or proliferate. It might be of interest to study the involvement of this dormant population in resistance to chemotherapy treatments.
The European Research Council (ERC) and the Fundación Botín and Banco Santander, through Santander Universidades, have financed this research.
Reference article:
Carme Cortina, Gemma Turon, Diana Stork, Xavier Hernando-Momblona, Marta Sevillano, Mònica Aguilera, Sébastien Tosi, Anna Merlos-Suárez, Camille Stephan-Otto Attolini, Elena Sancho & Eduard Batlle
A genome editing approach to study cancer stem cells in human tumors
EMBO Molecular Medicine (2017) DOI:10.15252/emmm.201707550
About IRB Barcelona
Created in 2005 by the Generalitat de Catalunya (Government of Catalonia) and University of Barcelona, IRB Barcelona is a Severo Ochoa Centre of Excellence, a seal that was awarded in 2011. The institute is devoted to conducting research of excellence in biomedicine and to transferring results to clinical practice, thus improving people’s quality of life, while simultaneously promoting the training of outstanding researchers, technology transfer, and public communication of science. Its 24 laboratories and seven core facilities address basic questions in biology and are orientated to diseases such as cancer, metastasis, Alzheimer’s, diabetes, and rare conditions. IRB Barcelona is an international centre that hosts more than 400 employees and 36 nationalities. It is located in the Barcelona Science Park. IRB Barcelona forms part of the Barcelona Institute of Science and Technology (BIST) and the “Xarxa de Centres de Recerca de Catalunya” (CERCA).
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).