Images
A bioinformatics analysis of pairing 64 drugs used to treat breast cancer allows researchers at IRB Barcelona to identify 10 previously untested combinations with potential to tackle resistance to breast cancer treatment.
In spite of the many drugs available to treat breast cancer, resistance continues to be a problem.
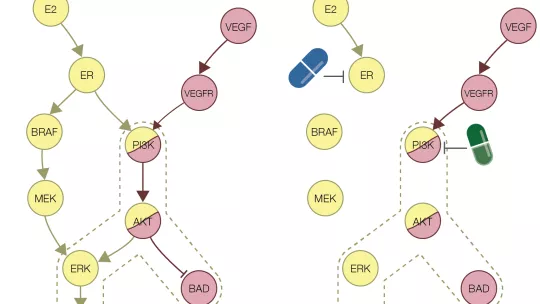
Based on the study of cell signalling networks, the cell signals that drugs alter when they reach their target molecule, an exhaustive in silico analysis of the pairing of 64 therapeutic agents used to treat breast cancer (half already in use and the other half in clinical testing phase) has allowed researchers at the Institute for Research in Biomedicine (IRB Barcelona) to identify 10 new and previously untested combinations that hold potential for the treatment of breast cancer.
Seven of the 10 combinations tested in breast tumour cells in vitro have shown a high level of synergy (the joint effect is greater than the sum of the individual effects) and one of these combinations has been validated in mice. The results in mouse models indicate that the combination of raloxifene and cabozantinib, two drugs prescribed by oncologists, “dramatically” boost the anti-tumour effects of each of the two drugs, as the authors explain in Cancer Research, the journal that has published the results in its advanced online edition.
Patrick Aloy, ICREA researcher and head of the Structural bioinformatics and network biology lab at IRB Barcelona, says, “we identify many more synergistic combinations in silico than combinatorial assays do until now with high-performance lab techniques, and we can provide experimental details. This implies that prior computational analyses give better results and are more reliable”. The researchers point out that in 70% of the combinations tested, the joint effects of the two drugs is “much much greater” than the effect of each alone, and therefore the same effect could be achieved with a smaller dose.
In this case, when scientists tested combined raloxifene and cabozantinib treatment in mice, they observed that the tumour shrank by 60%, while the individual effect of each drug merely prevented further tumour growth. Moreover, with the combination, a dose 3 and 25 times smaller, respectively, can be used compared to current treatments.
“This in itself is very important because drugs are in fact toxic and are used to kill cells. If, by using a smaller dose, a greater—or even the same—chemotherapeutic effect is achieved, it is a significant advantage with respect to reducing the side effects experienced by patients,” says Aloy. “Also, in principle, resistance would be avoided or delayed,” he comments.
When tumour cells learn to evade attack
In cancer treatment, including breast cancer, one of the problems faced by patients and oncologists is the onset of treatment resistance. Cancer cells become “insensitive” to the drugs that should kill them.
Resistance arises because the cancer cell, via the development of random mutations, learns to evade the effect of the drug. In 15% of cases, alternative molecular signalling pathways are activated to allow the tumour cells to divide again or to evade programmed cell death. Combined therapy using two or more drugs emerges as a promising approach to tackle this kind of resistance.
“Our analyses have allowed us to predict the signalling pathways that are inactivated by the joint action of two drugs,” explains Samira Jaeger, postdoctoral fellows and first author of the study. The scientists validated at the molecular level that the molecules predicted in the in silico model were indeed inhibited. “By combining drugs, we aim to attack the tumour cell simultaneously from various flanks, thus making it more difficult for the cell to resist treatment, as the pathways that allow it to survive and proliferate will be knocked out the same time,” she explains.
Future option
Having validated the computational network model, the scientists have three research lines running.
First, and with the aim to move towards clinical applications, they will test combined treatment with raloxifene and cabozantinib in tumours taken from patients and transplanted in mice. For this purpose, and as in the study just done, they will work in collaboration with ICREA researcher Angel R. Nebreda, a member of the Oncology Programme at IRB Barcelona.
Second, with the same aim to find more effective treatments for breast cancer, the lab will focus on pairing an anti-tumour agent and a drug administered for other conditions, such as those used to treat diabetes and high blood pressure.
Finally, Aloy’s lab is fine-tuning an experimental method that will allow them to validate the combined therapies that show the greatest long-term efficiency in treating resistance.
This study has been funded by the European Research Council (ERC Consolidator grant), the Ministry of Economy and Competitiveness and FEDER funds, a EU Marie Curie COFUND grant and the Generalitat de Catalunya.
Reference article:
Samira Jaeger, Ana Igea, Rodrigo Arroyo, Victor Alcalde, Begoña Canovas, Modesto Orozco, Angel R. Nebreda and Patrick Aloy
Quantification of pathway crosstalk reveals novel synergistic drug combinations for breast cancer
Cancer Research (2016) DOI: 10.1158/0008-5472.CAN-16-0097
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).