Images
The research group led by Travis Stracker identifies genes involved in male infertility in mice.
Their findings are relevant in humans and could explain undiagnosed cases of male infertility or subfertility.
Infertility affects about 15% of couples worldwide, and researchers estimate that about half of the cases are due to male infertility. “Despite a great deal of study, we still have a poor understanding of the origins of male infertility in many patients,” says Travis Stracker, head of the Genomic Instability and Cancer Laboratory at the Institute for Research in Biomedicine (IRB Barcelona).
Stracker and his team recently published a study in the journal Development that sheds light on the underlying causes of male infertility observed in patients with a rare ciliopathy called Reduced Generation of Multiple Motile Cilia (RGMC).
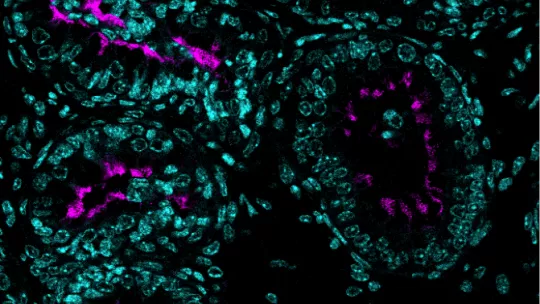
Patients with RGMC lack multiciliated cells, which are found in multiple tissues responsible for moving fluid, mucus or reproductive cells. Multiciliated cells have dozens of motile hairs, called cilia, that beat in a polarized and synchronized fashion to direct fluid flow across a tissue.
Stracker and his team studied mice lacking the genes GEMC1, MCIDAS or CCNO, which are important for multiciliated cell development. They found that male mice lacking one of these genes show a developmental defect in their reproductive system, specifically in the efferent ducts of the epididymis, a poorly understood structure that transports sperm from the testes to the vas deferens before ejaculation.
Although multiciliated cells had already been described in efferent ducts, it was not known that spermatozoid transport along the tubules relied on these specialized cells. The researchers have now demonstrated that a defect in or lack of multiciliated cells results in aggregates of spermatozoa that clog the ducts, preventing spermatozoa from moving from the testis to the epididymis, thereby causing infertility in mice.
Interestingly, these findings are easily translated to human pathologies. “Mutations in both MCIDAS and CCNO can cause RGMC disease in humans. “So, determining their role could help us to understand the cause of fertility issues in these patients,” explains Berta Terré, first author of the study. “Multiciliated cells in the efferent ducts are essential for male fertility and may be disrupted in other ciliopathies that could account for undiagnosed male infertility or subfertility in the population,” she explains.
This work has been supported by funding from the Ministry of Science, Innovation and Universities (previously known as MINECO), the Centres of Excellence Severo Ochoa Award and the Catalan Government.
Reference Article
Berta Terré, Michael Lewis, Gabriel Gil-Gómez, Zhiyuan Han, Hao Lu, Mònica Aguilera, Neus Prats, Sudipto Roy, Haotian Zhao, Travis H. Stracker
Defects in efferent duct multiciliogenesis underlie male infertility in GEM1, MCIDAS or CCNO deficient mice.
Development (2019). DOI:10.1242/dev.162628
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).