Images
Kennedy’s disease is an untreatable neuromuscular disorder considered a rare disease
A study led by Xavier Salvatella may pave the way for new lines of treatment for Kennedy’s disease
Kennedy’s disease is a rare neuromuscular disease caused by a mutation in the androgen receptor protein. The Laboratory of Molecular Biophysics led by Xavier Salvatella at the Institute for Research in Biomedicine (IRB Barcelona) has recently discovered new key structural features of the androgen receptor induced by the mutation. This finding has been published in the journal Nature Communications and could open up new therapeutic avenues for a family of incurable rare conditions called polyglutamine (polyQ) expansion diseases.
Spinal and bulbar muscular atrophy (SBMA), also known as Kennedy’s disease, is an untreatable neuromuscular disorder that causes a dramatic decrease in patients’ quality of life. It is classified as a rare disease, meaning that it affects less than 5 in 10,000 people. The mutation in the androgen receptor protein causing the disease is an expansion of a repetitive amino acid sequence containing only glutamine residues, called a polyQ tract. Interestingly, the length of this tract determines whether a person develops Kennedy’s disease.
A similar mutation can also be found in other polyQ expansion diseases, the most known of which is Huntington’s disease. Why an expansion of the polyQ tract leads to the development of polyQ expansion diseases in not understood. In the case of Kennedy’s disease, researchers believe that androgen receptor proteins containing a very long polyQ tend to oligomerize. These oligomers then generate toxic protein aggregates inside the cell, thereby impeding its proper functioning.
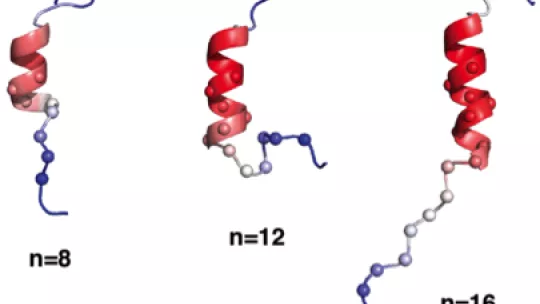
For a protein to function properly, it must fold into an ordered three-dimensional structure. Up to now, the part of the androgen receptor protein holding the polyQ expansion was thought to be disordered and therefore unable to fold into such a structure. In 2016, Salvatella’s group found that the polyQ tract of the androgen receptor could adopt a helical structure. “This was a very interesting result, as so far these tracts were thought not to adopt any specific structure,” explains Salvatella.
In the current study, the team went one step further and visualized the polyQ tract protein helix via advanced biophysical and computational methods. They now report that this specific region can form well-ordered helices and, crucially, that the polyQ expansion greatly increases the degree of order and stability of these helices. This information furthers our understanding of androgen receptor function.
Albert Escobedo, postdoctoral researcher at IRB Barcelona and co-first author of the study, says, “Our findings indicate that mutated long polyQ tracts can form increasingly stable helices. This suggests that they may cause polyQ disorders, such as Kennedy’s disease.” He emphasizes that “targeting these helices with drugs may be a therapeutic avenue for these currently incurable disorders.”
This study was funded by grants from the Marató de TV3, the European Research Council (ERC), and the Ministry of Science, Innovation and Universities (previously known as MINECO).
Reference article:
Albert Escobedo, Busra Topal, Micha B. A. Kunze, Juan Aranda, Giulio Chiesa, Daniele Mungianu, Ganeko Bernardo-Seisdedos, Bahareh Eftekharzadeh, Margarida Gairí, Roberta Pierattelli, Isabella C. Felli, Tammo Diercks, Oscar Millet, Jesús García, Modesto Orozco, Ramon Crehuet, Kresten Lindorff-Larsen and Xavier Salvatella.
Side chain to main chain hydrogen bonds stabilize a polyglutamine helix in a transcription factor.
Nature Communications (2019) DOI: 10.1038/s41467-019-09923-2
About IRB Barcelona
The Institute for Research in Biomedicine (IRB Barcelona) pursues a society free of disease. To this end, it conducts multidisciplinary research of excellence to cure cancer and other diseases linked to ageing. It establishes technology transfer agreements with the pharmaceutical industry and major hospitals to bring research results closer to society, and organises a range of science outreach activities to engage the public in an open dialogue. IRB Barcelona is an international centre that hosts 400 researchers and more than 30 nationalities. Recognised as a Severo Ochoa Centre of Excellence since 2011, IRB Barcelona is a CERCA centre and member of the Barcelona Institute of Science and Technology (BIST).