ICREA Research Professor, ERC Starting Grant

Meet Our Scientists Videos
Research information
Background
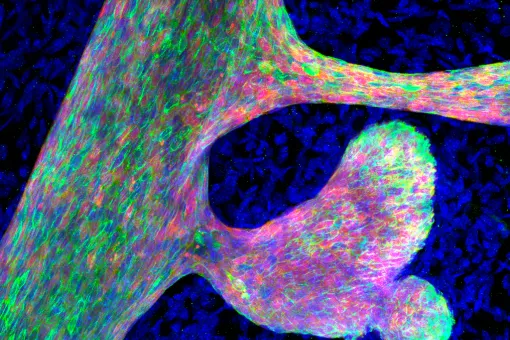
Adult stem cells ensure maintenance of the tissue and integrity in response to damage. They self-renew, are multipotential, and respond to and repair injuries. The skin and other stratified epithelia as model tissues display a high turnover rate, and contain well-defined populations of adult stem cells (SCs). A key feature of SCs is that they localize to specialized niches, where they remain unspecified but sense the needs of the tissue. Constant tissue replenishment drives a SCs to proliferate, the daughter cells to egress the niche, propagate, and terminally differentiate. Niche adherence, dormancy, activation, and exit, must be finely regulated processes, since loss of this tight control is causative of aging, and the onset and progression of squamous cell carcinomas.
Research interests
Our global aim is to characterize the molecular pathways that regulate the behaviour of normal stem cells during homeostasis, and how they become deregulated during carcinogenesis and ageing.
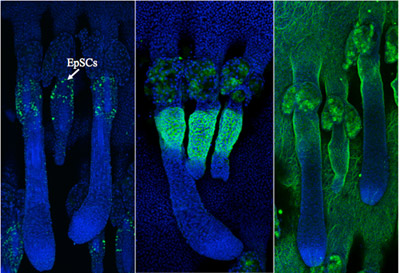
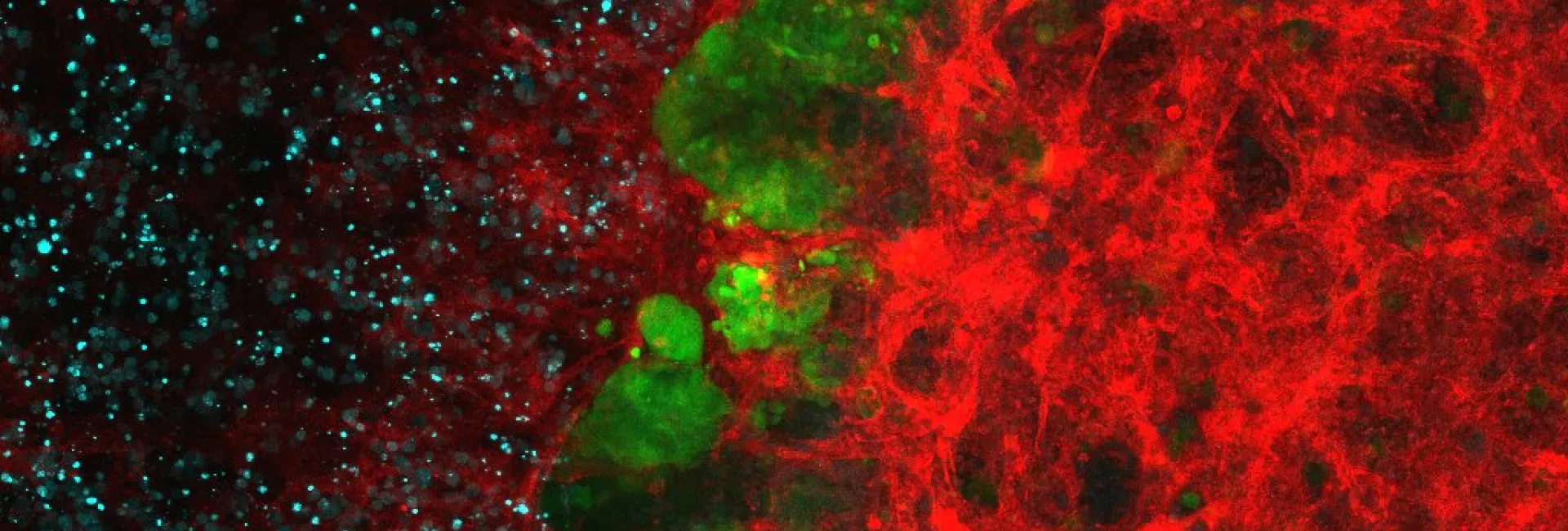
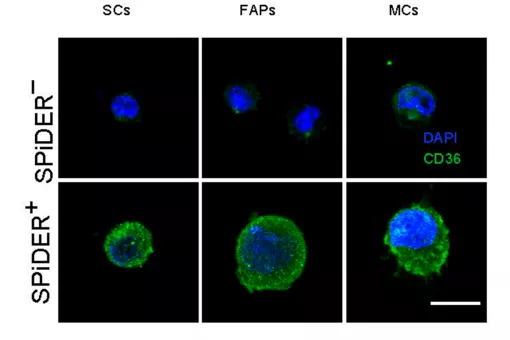
Epidermal stem cells from mouse tail skin visualized by wholemount 3D immunostaining: Left panel shows epidermal stem cells marked with BrDU (Label Retaining Cells); Middle and right panels Show expression of keratin-15 and high expression of integrin alpha6, respectively.
Research lines
We are interested in studying the following questions:
- What intrinsic (cell autonomous) mechanisms regulate adult stem cell function, and how are they deregulated during ageing and cancer?
- How do adult stem cells communicate with their local environment (i.e. niche) and how is the system coordinated to dictate stem cell function?
- What systemic cues communicate with adult stem cells and how do distant adult stem cells either from the same tissue or from different tissues coordinate their function at the level of the whole organism?
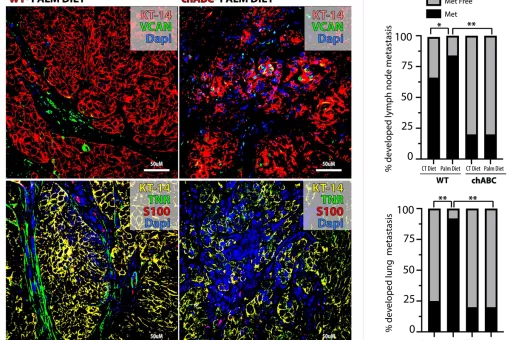
- How does deregulation of pathways that control stem cell behaviour contribute to the maintenance and invasive progression of skin and oral squamous cell carcinomas (SCC)? Is there cancer stem cell heterogeneity in human SCC? If so, what is its significance?
We use a combination of conditional mouse models, primary cell culture (human and mouse), live microscopy, highthroughput genomics and proteomics approaches, and basic molecular biology.
Selected publications
Projects
"Mecanismo de acción e inhibicion terapéutica de la población de celulas stem de la metástasis sensibles a la dieta" Ministerio de Economía y Competitividad and Fondo Europeo de Desarrollo Regional (FEDER-UE). Reference: BFU2016-79242-P


Grup de Recerca consolidat (SGR 2017-2019) Secretaria d'Universitats i Recerca del Departament d'Empresa i Coneixement de la Generalitat de Catalunya. Agencia de Gestió d'Ajuts Universitaris i de Recerca (AGAUR). Reference: 2017 SGR 1109


"Studying and targeting diet-responsive metastasis initiating cells" Worldwide Cancer Research (WCR). Referencia: 18-0133

"Células madre y cáncer" Fundación Botin (Banco Santander)

"Dietary Influences on Metastasis: How, When, and Why" (LIPOMET) European Research Council (ERC) under Horizon 2020 Programme. Reference: 787041

"Identificación de nuevas estrategias terapéuticas contra las células iniciadoras de la metástasis". Fundación Cientifica de la Asociación Española contra el Cáncer (FCAECC). Reference: PROYE19006AZNA

"Àcids grassos de la dieta i metàstasi: identificació de noves estratègies terapèutiques contra les cèl·lules iniciadores de la metàstasi" funded by Fundació La Marató TV3. Reference: 201902-30
"Impacto de nuestra dieta sobre el proceso de la metástasis tumoral" funded by the Agencia Estatal de Investigación. Reference: PID2019-107583GB-I00 / AEI / 10.13039/501100011033
"Nueva inmunoterapia contra el cáncer: bloqueo de la reprogramación lipídica basada en el mapeo de transcriptomas de metástasis (LipIMMUNE)" funded by the project PLEC2021-007654, funded by MCIN/AEI/10.13039/501100011033 and by the European Union “NextGenerationEU”/PRTR”